 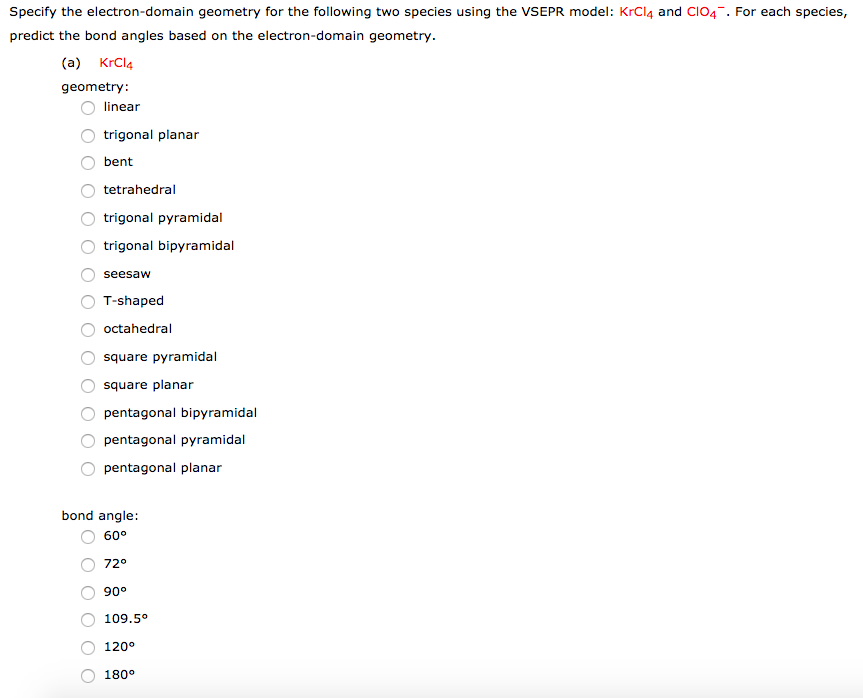
c) CH3I is tetrahedral, but not symmetrical, making us uncertain of. The difference with a polar covalent bond is 0.4 to 1.8 and a nonpolar covalent bond is from 0 to 0.4. b) BCl3 is trigonal planar and symmetrical, making us confident the bond angles are 120. 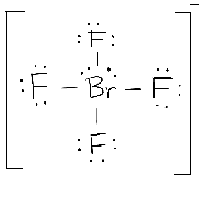
If the difference of the electronegativity between the two elements is greater than 1.7 then the bond is ionic. So, that bends the axial fluorines together a bit. Predict if a bond between two atoms is nonpolar covalent, polar covalent, or ionic. Note though that the structure is distorted a bit due to the repulsive forces of the lone pair of electrons you see (not bonded). With one lone pair of valence electrons, you get a seesaw molecular geometry. The base angles are still 180, 120, and 90 while the tweaked angle will now be slightly less in each case due to the extra. 
If all the bonds are in place the shape is also trigonal bipyramidal. SF4Seesaw Molecular Geometry - The seesaw shape maximises the bond angles of. The T shape and trigonal bipyramidal geometry of iodine trichloride lead to a bond angle (Cl-I-Cl) slightly smaller than 90 ° and greater than. SF4 Molecular Geometry / Shape Wayne Breslyn 634K subscribers Subscribe 352. Therefore, you can put 6x4 on each fluorine, 2x4 to account for four single bonds, and 2 for the last 2 valence electrons available.Īs a result, you have 5 electron groups, so the electron geometry would be trigonal bipyramidal. With one lone pair on this electronic geometry, the lone pair must occupy an equatorial position and the molecular geometry becomes see-saw shape. For sp3d hybridized central atoms the only possible molecular geometry is trigonal bipyramidal. The molecular geometry and shape of the molecule can be predicted from the following table: General formula: Number of bond pairs (B) Number of lone pairs (E) Molecular geometry: Shape: AX: 1: 0. A molecule with seesaw molecular geometry has a bond angle of less than 90 and 120. When a lone pair is added, it is placed as far away as possible from the bonding pairs due to electron-electron repulsion. You can put sulfur in the middle because fluorine tends to make single bonds. In Seesaw molecular geometry where there are four bonds attached to a central atom and one lone pair. Within the context of VSEPR theory, you can count electrons to determine the electron geometry ("parent" geometry). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
